Clinical Trials GPS Launches Nationwide Patient Recruitment Site; Contains Over 25,000 Trials and 15,000 Facilities Across the U.S.
Clinical Trials GPS was created to provide individuals with the most comprehensive, up to date information about ongoing clinical drug trials, as well as a quick and easy way to participate.
Visitors to the site can search for the latest clinical trials in their area, browse through the directory of clinical trial facilities, read educational articles to learn more about the various conditions treated by clinical trials, or speak with a clinical trials expert at (877) 330-2919.
The site has a comprehensive listing of all open trials in the United States and is categorized to help visitors find them quick. Clinical Trials GPS has information on trials from all the leading pharmaceutical companies, academic institutions and trials ranging from Cancer to Diabetes and Vaccines to Osteoarthritis.
“The company’s mission is ultimately to assist people in finding solutions to their health problems”, says John Longan, DIrector of Business Development at Clinical Trials GPS.
“Clinical trials are a critical part of the drug development process and all drug treatments must go through an FDA monitored, rigorous process. Every drug in the United States has been through this process and all new drug innovations must obtain FDA approval. Clinical Trials GPS was developed to guide people through the process of learning about clinical trials, help people find a trial that may be of interest, and connect them with the closest research site.”
ClinicalTrialsGPS.com
The new site contains some important features like a powerful search engine, interactive maps, and a personalized dashboard. This amazing technology is also backed by an industry leading call center, equipped to help guide patients through the process of finding the right trials and the right facility for their needs.
Visitors to the site can find a trial in one of 3 ways:
- Select the condition(s) that are relevant to the user
- Enter their zip code to find local trials, or
- Call directly into the call center to speak with a Clinical Trials Expert
Once a visitor completes a short form they are forwarded to a personalized dashboard, which suggests related trials, facilities and informational articles related to their condition.
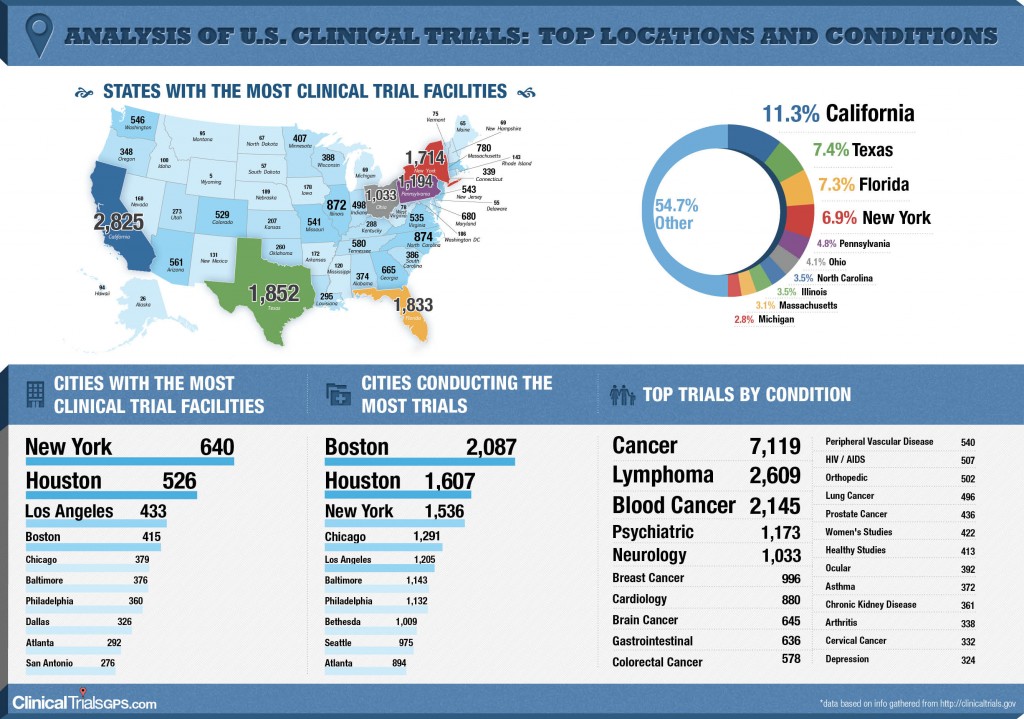
Analysis of U.S. Clinical Trials; Top Locations and Conditions (INFOGRAPHIC)
Recently, the CTGPS team created a graphical representation of the data found on the Clinical Trials GPS website (which is derived from publically available data found at clinicaltrials.gov). It includes an analysis of the:
- States with the most Clinical Trial Facilities
- Cities with most Clinical Trial Facilities
- Cities conducting the most Trials
- Top Trials by Condition
One of the most interesting discoveries was that 6 of the top 10 conditions that trials are being conducted for are cancer related.
Clinical Research Sites
One of the biggest struggles that clinical research sites have includes recruiting patients to meet enrollment goals. Working with Clinical Trials GPS is another part of your recruitment strategy. In addition to listing your trials on Clinical Trials GPS, we can work with you to highlight your trials to our visitors, create site profile pages, and even assist with other online Search Engine Marketing campaigns at an affordable cost. Clinical Trials GPS can connect you with qualified prospective participants for any indication trial as part of your overall marketing strategy. With experience in both online and offline marketing, contact us today to learn how we can help you recruit for your trials.
Pharmaceutical and CRO Sponsors
Clinical Trials GPS is another great avenue to promote your open or upcoming trials. With a comprehensive listing of trials across the US, we are able to help you find subjects for your trials at any location. In addition to sending email blasts to interested participants, we can also create personal online campaigns ranging from trial-specific pages to trial-specific micro-sites and even execute Google AdWords campaigns to help ensure traffic from all interested web searchers. For more information, contact: John Longan (877) 248‑5589